|
Therefore, the radioactive decay constant represents the fraction of radioactive atoms that disintegrates in unit time. The quantity dN/N represents the fraction of the total number of atoms that disintegrate in time dt. The radioactive decay formula is given below the picture,įrom the above formula, – (dN/N)/dt = k = rate constant.  
If N 0 number of the atoms present at the time t = 0 and N number of atoms present after the time t. Therefore, dN/dt = – kN, where N = number of the atoms of the disintegrating radioactive element, dt = time over which the decay is measured, and k = radioactive decay constant. The rate of radioactive decay formula depends only on the single power of the radioactivity. 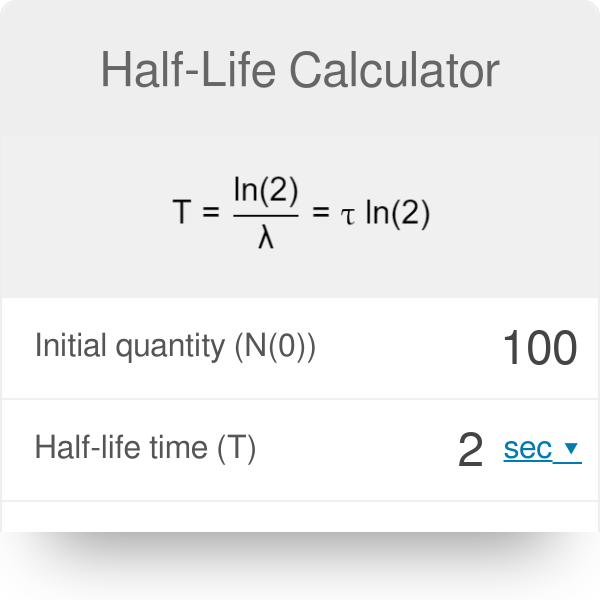 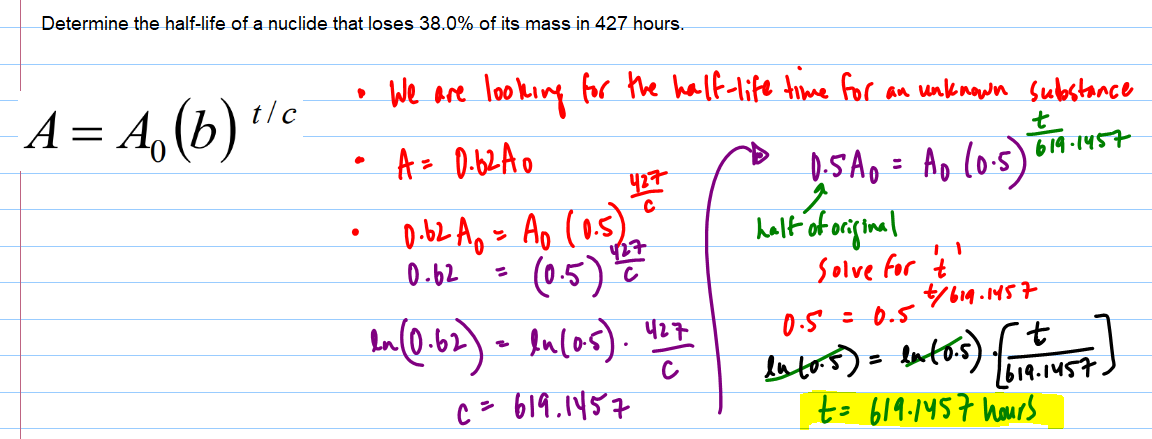
It is used mainly in radiocarbon dating and the determination of the age of matter or materials. Radioactive decay is one important natural phenomenon obeying the first-order chemical kinetics.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
